 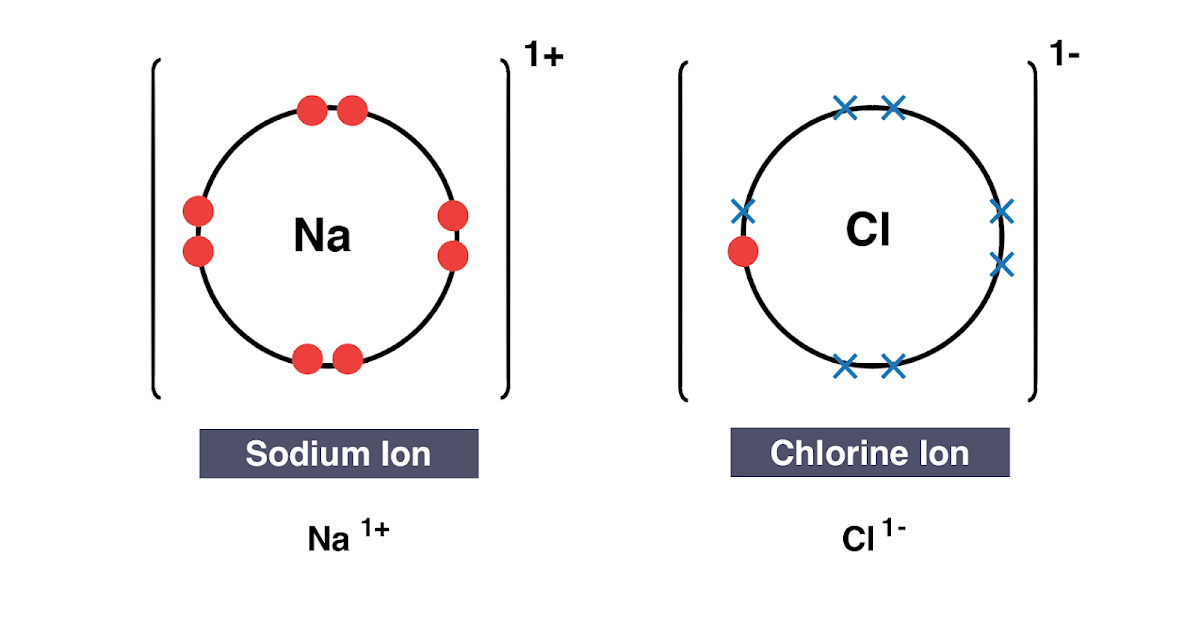
Non-metal atoms gain electrons to become negatively charged ions.Metal atoms lose electrons to become positively charged ions.When a metal atom reacts with a non-metal atom electrons in the outer shell of the metal atom are transferred.4.2.1 Chemical bonds, ionic, covalent and metallic.4.2 Bonding, structure and the properties of matter.know that ionic bonding is the strong electrostatic attraction between oppositely charged ions Ionic bonding involves electrostatic attraction between oppositely charged ions in a lattice. 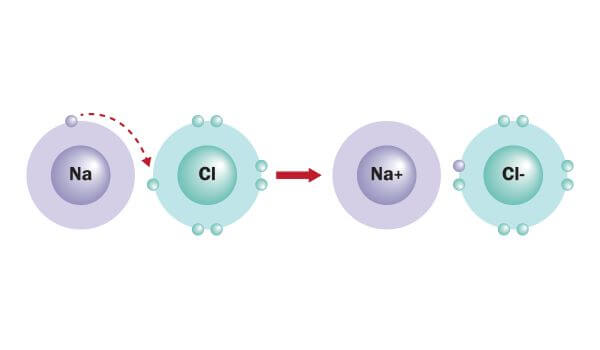
a) ionic bonding as electrostatic attraction between positive and negative ions, and the construction of 'dot-and-cross' diagrams.Examination of a model of the NaCl crystal.Representation of ionic bonds using dot and cross diagrams.1.2.4 demonstrate knowledge and understanding that: ionic bonding involves attraction between oppositely charged ions ionic bonds are strong and substantial energy is required to break ionic bonds. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
